Radium at its core
Brief overview: ø
State of matter: solid
Metal
atomic mass: 226
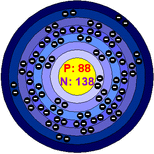
atomic number:88
Number of protons: 88
Number of neutrons: 138
Number of electrons:88
Radium was discovered by Marie Curie in 1898
Density (at 20 degrees C) : 5.5 g/cm3
State of matter: solid
Metal
atomic mass: 226
atomic number:88
Number of protons: 88
Number of neutrons: 138
Number of electrons:88
Radium was discovered by Marie Curie in 1898
Density (at 20 degrees C) : 5.5 g/cm3
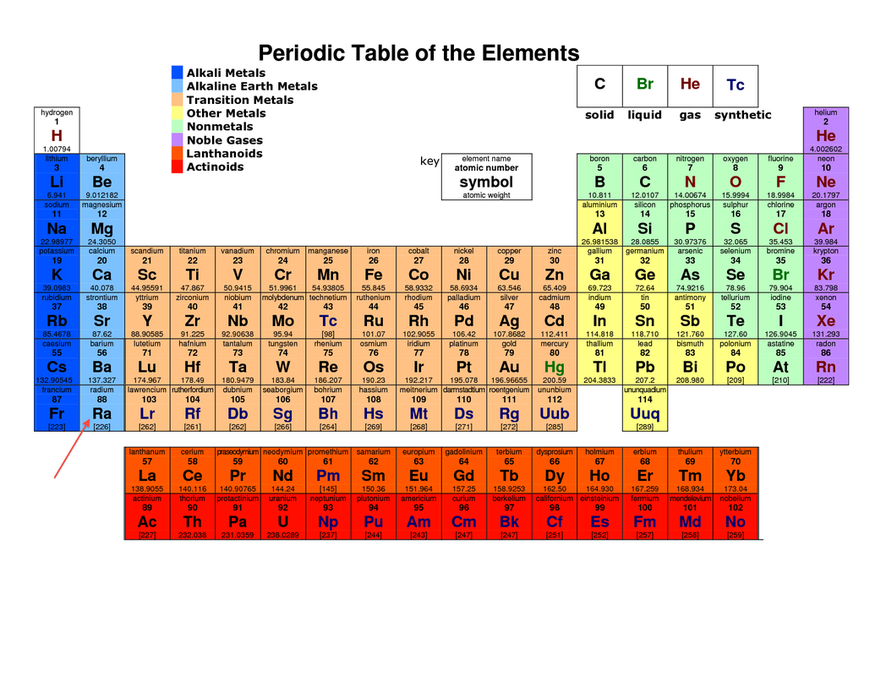
Radium on the periodic table!
Radium is located in the 2nd group, in the 7th period
© http://www.epa.gov/radiation/radionuclides/radium.html
øhttp://www.chemicool.com/elements/radium.html
˚http://www.endmemo.com/chem/common/radium.php
øhttp://www.chemicool.com/elements/radium.html
˚http://www.endmemo.com/chem/common/radium.php